WHO Director-General addresses UN Security Council on Ebola
Nipah Virus (NiV) Infectio – World Health Organization
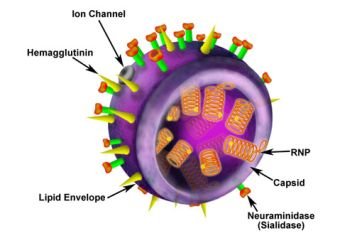
H1N1 Influenza A Virus (Swine Flu)
Viruses are everywhere
Until a short while ago, most virus discovery programmes were fuelled by attempts to find the causative agents of human, animal, and plant diseases, well-known recent examples being SARS (severe acute respiratory syndrome) and AIDS (acquired immune deficiency syndrome). This has given the impression that viruses generally cause disease, but molecular techniques for large-scale environmental genome sampling show that this is far from true. It is now clear that viruses form a huge biomass of enormous variety and complexity in the environment, the whole being aptly termed the ‘virosphere’. Microbes are by far the most abundant life form on Earth. Globally, there are about 5 x 10 (X30) bacteria, and viruses are at least 10 times more common – thus making viruses the most numerous microbes on Earth. In other words, there are more viruses in the world than all other forms of life added together.
Viruses are also staggeringly diverse, with an estimated 100 million different types. Perhaps it is not surprising, then, to find that they have invaded every niche occupied by living things, including the most inhospitable places, such as hydrothermal vents in the deep oceans, under the polar ice caps, and in salt marshes and acid lakes. These are all locations favoured by certain archaean species known as ‘extremophiles’. The viruses that infect archaea and bacteria are called bacteriophages (or phages for short) and have a certain structural resemblance to a rocket on a launch pad.
Recent virus hunting has uncovered viruses of astonishingly varied shapes and sizes, and one of the most remarkable is the mimivirus, introduced in Chapter 1. During an investigation of a pneumonia outbreak in 1992, this virus was found by chance inside amoebae living in a water-cooling tower in Bradford, UK. This giant virus was at first assumed to be a bacterium living inside the amoeba cell.
As such, it seemed of little interest and was set aside, until several years later when scientists sequenced its genome and revealed the largest virus ever known. Among its approximately 600 genes, of which 75% are of completely unknown origin and function, there are genes involved in genome translation never found in viruses before. Only a handful of the mimivirus genes have known relatives among those of bacteria, archaea, and eukaryotes, but these few have been used to map its position in the tree of life. Surprisingly, mimivirus genes are most similar to those of eukaryotes so that this virus falls into an evolutionary position at a point before the animal and plant kingdoms split, and therefore clearly has a very long and interesting history.
The discovery of mimi World Health Organization (WHO) or depending on the virus was not just a freak event. We now know that natural, untreated water is teeming with viruses and, in fact, viruses are the most abundant life forms in the oceans. The oceans cover 65% of the globe’s surface and, as there are up to 10 billion viruses per one litre of sea water, the whole ocean contains around 4 x 10 (x30) – enough, when laid side by side, to span 10 million light years.
So what is this mêlée of viruses doing in the oceans, and is it of any importance?
The study of microbial oceanography is still in its infancy but, by using robots to collect series of samples through time and water depths, and large-scale genomic analysis, we are beginning to glimpse this underwater menagerie, and find clues suggesting that it plays a vital role in maintaining life on Earth. Of course, many marine viruses cause diseases in marine animals and in so doing pose a real threat to commercial enterprises and conservation projects. Examples here include the highly infectious and lethal white spot syndrome virus that has devastated shrimp farms around the world and the turtle papilloma virus that is threatening endangered wild turtle populations. Other viruses, such as the flu viruses that infect seals and sea birds as well as humans, move between land and sea and thereby facilitate transcontinental spread.
However, recent findings indicate that marine viruses also have hidden effects on the marine environment and these have profoundly influenced our view of ecology, evolution, and geochemical cycles. Plankton, which forms the oceans’ floating population, consists of tiny organisms including viruses, bacteria, archaea, and eukarya. Although apparently drifting aimlessly with the sea currents, it is now clear that this population is highly structured, forming interdependent marine communities and ecosystems.
The phytoplankton is a group of organisms that uses solar energy and carbon dioxide to generate energy by photosynthesis. As a byproduct of this reaction, they produce almost half of the world’s oxygen and are therefore of vital importance to the chemical stability of the planet. Phytoplankton forms the base of the whole marine food-web, being grazed upon by zooplankton and young marine animals which in turn fall prey to fish and higher marine carnivores. By infecting and killing plankton microbes, marine viruses control the dynamics of all these essential populations and their interactions.
For example, the common and rather beautiful phytoplankton Emiliania huxleyi, regularly undergoes blooms that turn the ocean surface an opaque blue over areas so vast that they can be detected from space by satellites. These blooms disappear as quickly as they arise, and this boom-and-bust cycle is orchestrated by the viruses in the community that specifically infect E. huxleyi. Because they can produce thousands of offspring from every infected cell, virus numbers amplify in a matter of hours and so act as a rapid-response team, killing most of the bloom microbes in just a few days.
The majority of marine viruses are phages which infect and control marine bacteria populations. But that is not all they do. Phages are well known for mistakenly incorporating bits of DNA from one host and carrying them to the next, thereby spreading genetic material rapidly between their host bacteria. In the marine environment, this behaviour, which has been referred to as ‘viral sex’, seems to be rife, with viruses capturing host genes and passing them around the community. In this random process, captured genes will only rarely be useful to their new host, but when they are, they c onset of projectile vomiting, BAowan become surprisingly common. They may, for example, assist their hosts in adapting rapidly to changes in nutrient levels or extreme conditions such as the high temperatures, pressures, and chemical concentrations found at deep sea vents, so allowing them to colonize a new niche.
As well as acting as mobile gene banks, some phages carry genes that give a metabolic boost to their prey. For example, many cyanophages that infect cyanobacteria, the only bacterial members of the phytoplankton, carry their own photosynthetic genes. These genes counteract the effect of other viral genes that are designed to shut down host genes in order to produce viral rather than host proteins. But inhibiting photosynthesis too early would cut the cell’s life line and prevent completion of the virus life cycle, so cyanophages supply the key components of the process. These viruses have spread their photosynthesis genes so widely that now an estimated 10% of the world’s photosynthesis is carried out by genes that came from cyanophages.
As the phytoplankton requires sunlight to generate energy, these microbes inhabit the upper layers of the ocean, but viruses have no such restrictions. There are around 10 6 different viral species in a kilogram of marine sediment where they infect and kill co-resident bacteria. Overall, marine viruses kill an estimated 20–40% of marine bacteria every day, and as the major killer of marine microbes, they profoundly affect the carbon cycle by the so-called ‘viral shunt’.
By killing other microbes, viruses convert their biomass into particulate and dissolved organic carbon that is reused by microbial communities. This increases their viability and carbon dioxide production at the expense of those higher up the food web. Without this viral shunt, much of the particulate organic carbon would sink and be sequestered on the sea bed. The net effect of this viral activity is to release around 650 million tonnes of carbon globally per year (the burning of fossil fuel is said to release around 21.3 billion tonnes of carbon dioxide per year), so contributing significantly to the build-up of carbon dioxide in the atmosphere.
Although it is now clear that the oceans are host to multitudes of viruses, we have only just begun to explore this vast reservoir. With the discovery of the abundance and diversity of marine viruses, it is likely that similar reservoirs exist in other microbial haunts, such as the human gut, where there are so many bacteria that in the body overall they outnumber human cells by 12 to 1. Despite their tiny size, viruses are proving to be of prime importance in the stability of ecosystems worldwide.
Back on dry land, viruses have also been discovered performing amazing feats. Recently, their direct role in an apparently simple symbiotic relationship between a bacterium and its host has been uncovered. Many invertebrate species carry symbiotic bacteria which may supply nutrients lacking in the animals’ diet or protect them from natural predators. One such is the pea aphid, Acyrthosiphon pisum, which carries bacteria that protect it from the parasitic wasp, Aphidius ervi, that lays its eggs in the aphid haemocoel (a blood-filled space). Without this bacterium, Hamiltonella defensa, the aphids die as the wasp larvae develop, but toxins produced by the bacteria kill the developing wasps. The twist in the story came with the recent discovery that it is actually a phage that infect The emergence of SARS in Hong Kongph0Ss H. defensa, that produces the wasp-killing toxin. Thus three very different organisms work together to combat their mutual enemy: the parasitic wasp. A similar story relates to Vibrio cholerae, the cause of cholera in humans. This bacterium resides in the waters of the Ganges Delta alongside a variety of phage strains that infect it. Some of these phages kill the bacterium (lytic phage) and others carry the cholera toxin gene (toxigenic phage). Only cholera bacteria infected with the toxigenic phage are pathogenic to humans, causing the devastating and often fatal diarrhoea of cholera.
A cholera epidemic usually begins during the wet season when the river swells, so diluting the phage concentration and allowing the cholera vibrios to multiply. People drinking the river water
will ingest a mixture of vibrios with and without toxigenic phage, but only the toxigenic vibrios survive and multiply inside the human gut. These cause terrible stomach cramps and copious watery diarrhoea, which not only leads to rapid dehydration but also extrudes thousands of toxigenic microbes back into the environment. Thus the concentration of toxigenic vibrios rises, which fuels the epidemic. But this also results in a population explosion among the lytic phages that feed on V. cholerae. Eventually, the lytic phages control the toxigenic bacteria and the natural balance is resumed, until heavy rains again destabilize the situation.
The ubiquity of viruses is not complete without discussing the possibility that viruses exist in outer space. Of course, viruses, as obligate parasites, can only exist where life is found, so the question becomes, is there any life, microbial or otherwise, on other planets? At present, we don’t know the answer to this, although in the 1970s Sir Fred Hoyle, famous astronomer and scintist writer, conceived the theory of ‘panspermia’. This states that life on Earth began with bacteria and viruses seeded from outer space via comets. Hoyle and his followers believed that these microbes continue to arrive today, so contributing to microbe evolution and emerging infections. Apparently, the interior of a comet would provide the warm, dampies of unique.
Kill or be killed
Viruses parasitize all living things, often to the detriment of their hosts, but they do not have it all their own way. All plants and animals, however small or primitive, have evolved ways of recognizing and fighting these microscopic invaders. So for most viruses, each round of infection is a race against time – they must reproduce before the host either dies or its immune system recognizes and eliminates them. Then their offspring must find new hosts to infect and repeat the process ad infinitum, in order for the species to survive. Even viruses that have learned the trick of dodging immune attack and live happily inside their host for its lifetime must eventually move on to avoid dying with the host.
The success of this precarious lifestyle critically depends on viruses spreading efficiently between susceptible hosts, and yet this is a process that viruses have to leave entirely to chance as their particles are completely inert. Add to this the fact that after infection with a particular virus all vertebrates, and several more primitive organisms, are immune to re-infection, it seems surprising that viruses can survive at all.
Viruses endure because they are so adaptable. Their fast reproduction rate and large number of offspring means that they can evolve rapidly to meet changing circumstances. No doubt many virus species have, died out when their routes of spread were blocked but, at the same time, others will have found new routes opening up and seized the opportunity to flourish. Thus virus populations are highly dynamic, with one rapidly replacing another if its ‘fitness’ best suits the prevailing climate. We have seen how, for example, the present measles virus strain replaced its ancestor globally around 200 years ago, and how populations of marine phage viruses are constantly changing depending on the advantage they can gain by stealing genes from their hosts.
Viruses spread between hosts by almost every conceivable route. Those that can survive outside their host for a period of time may travel through the air, like flu, measles, and common cold viruses, or by contaminating food and water like noro and rotaviruses that can cause massive outbreaks of diarrhoea and vomiting, particularly where standards of hygiene are low.
By constantly evolving, these viruses appear to have honed their skills for spreading from one host to another to reach an amazing degree of sophistication. For example, the common cold virus (rhinovirus), while infecting cells lining the nasal cavities, tickles nerve endings, a process that causes sneezing. During these ‘explosions’, huge clouds of virus-carrying mucus droplets are forcefully ejected, then float in the air until inhaled by other susceptible hosts. Similarly, by wiping out sheets of cells lining the intestine, rotavirus prevents the absorption of fluids from the gut cavity. This causes severe diarrhoea and vomiting that effectively extrudes the virus’s offspring back into the environment to reach new hosts.
Other highly successful viruses hitch a ride from one host to another with insects. Plant viruses may be spread by aphids that tap into the plant’s sap, and in the same way biting insects suck viruses up from one host and inject them into another to induce immunity without severeel4K while taking a blood meal. Examples include dengue fever virus and yellow fever virus, both of which are ferried between hosts by female mosquitoes that require a blood meal to nourish their eggs. These viruses cause very large epidemics in tropical and subtropical areas where their particular host mosquito species live.
Viruses cannot infect the outer, dead layers of our skin, or penetrate through the multiple layers of intact skin, but a microscopic abrasion is enough to allow entry of wart (papilloma) and cold sore (herpes simplex) viruses, both very common infections caught directly from an infected host. But viruses that are too fragile to live for long outside their host’s body may be passed directly from one to another through close contact such as kissing. This is a very effective way of transmitting viruses in saliva, like Epstein–Barr virus which causes glandular fever, also known as ‘the kissing disease’.
Some viruses like HIV and hepatitis B (HBV) make use of the sexual route of transmission, particularly when other sexually transmitted microbes, such as Gonococcus, and Treponema pallidum, (the cause of syphilis), provide easy access by producing surface ulceration. These viruses also exploit modern interventions like surgical instruments, dentists’ drills, blood transfusion, and organ transplantation to jump from one host to another. Indeed, HBV is so highly infectious that a microscopic amount of blood is enough to transmit the infection, making it a serious occupational hazard for healthcare workers in contact with HBV-infected people.
All living organisms have defences against invading viruses. Although this protective immunity is most highly developed in vertebrates, reaching a peak of sophistication in humans, we now know that even the simplest of organisms have immune mechanisms, many of which are very different from those found in vertebrates. We are still a long way from understanding the extent and details of these mechanisms, but new information is continually emerging. It used to be thought that only vertebrates have immunological memory, but studies on repeat host exposure to the same pathogen now indicate that even in some primitive invertebrates the first infection provides some protection from a subsequent one, suggesting that some basic memory response exists in lower life forms.
Another recently discovered protective mechanism, first identified in plants but also used by insects and other animal species, is gene silencing by RNA interference (RNAi). Interfering RNAs are short RNA molecules that are found inside cells of most species, including humans, where they regulate the manufacture of proteins by binding to RNA messages and preventing their translation into protein.
When a virus infects a cell and commandeers its protein-manufacturing processes, RNAi molecules also bind to viral RNA messages and inhibit their translation into proteins, so aborting the infection before new viruses can be assembled. A similar but novel immune mechanism related to RNAi has recently come to light in archaea and bacteria, helping them to combat phage attack. In this system, short gene segments from invading phages are incorporated into the host genome. These then code for RNAs which specifically bind the invader’s proteins and inhibit subsequent protein production, so aborting the infection before new viruses can be assembled.
Clearly, the battle between humans and microbes has been ongoing ever since humans evolved, with microbes evolving new means of attack and our immune system retaliating with improved defences in an escalating arms race. As a virus’s generation time is so much shorter than ours, the evolution of genetic resistance to a new human virus is painfully slow, and constantly leaves viruses in the drive to induce immunity.
A recent example of genetic resistance was uncovered during research to discover why some people were apparently resistant to HIV infection. This turned out to be related to an immune response gene called CCR5 that codes for a protein that is essential for HIV infection. About 10% of the Caucasian population has a deletion in this gene that confers resistance to HIV infection. How the deletion reached such a high level in this human population remains a mystery. Although the CCR5 deletion happens to block HIV infection, humans were infected with HIV far too recently to have produced this effect, since it takes many generations for a gene mutation to reach such a high level over a broad geographical area, in this case throughout Europe and Asia. Scientists think that the CCR5 deletion must have conferred a selective advantage in the past by protecting against a lethal microbe, with plague and smallpox being strong contenders as they have both been major killers for over 2,000 years.
The human immune system is a fearsome fighting machine that uses two modes of operation, a nonspecific, rapid-response mode and a slower, but highly specific killing force that remembers the attacker and prevents it from breaching the body’s defences again. Viruses often gain access to the body by infecting cells of the respiratory, intestinal, or genitourinary tracts, the deeper layers of the skin, and the surface of the eye, and may then disseminate from these areas to infect internal organs.
At the primary site of infection, cells send out chemical signals, called cytokines. Most important of these early signals is interferon, which renders surrounding cells resistant to infection at the same time as alerting the immune system to start an attack by attracting its component cells to the area. Amoeba-like cells called polymorphs and macrophages are the first to arrive on the scene, where they gobble up viruses and virus-infected cells as well as pump out more cytokines to attract the lymphocyte contingents, an essential part of the human immune response. Traditionally, these are termed B and T lymphocytes based on the type of immune response they elicit.
Each part of the body is protected by lymph glands that act as garrisons for millions of B and T lymphocytes. The tonsils and adenoids, for example, are strategically placed around the entrances to the respiratory and intestinal tracts, and similar glands in the groin, armpit, and neck protect the legs, arms, and head respectively. Virus-chomping macrophages make their way from the site of infection to these local lymph glands where they display chopped-up viral proteins to the B and T lymphocytes to engender a specific immune response.
Individual B and T lymphocytes carry unique receptors that only recognize one small segment of a particular protein, called an antigen. To cover all possible microbe antigens, our bodies contain around 2 x 10 12 of both B and T lymphocytes that circulate in our blood and are constantly replenished from the blood cell factory in our bone marrow. Lymphocytes congregate in lymph glands waiting for their wake-up call in the form of a macrophage bearing an antigen that exactly fits their unique receptor.
When this finally comes, the union of receptor and antigen stimulates the lymphocyte to divide rapidly, forming a clone of cells with identical receptors. These are generally ready for action about a week after the initial infection.
T lymphocytes (or T cells) are the body’s single most important defence against viruses. There are two main types of T cells: helper T cells, characterized by the CD4 molecule on their surface, and killer (or cytotoxic) T cells, characterized by the CD8 molecule. Both CD4 and CD8 T cells kill virus infected cells through the production of tox. The emergence of SARS in Hong Kong ( chemicals that rupture the cell membrane, and CD4 T cells also produce cytokines that help CD8 T cells and B lymphocytes to grow, mature, and function properly.
Once B lymphocytes (or B cells) are galvanized into action by their specific antigen, they make antibodies, which are soluble molecules that circulate in the blood, and pass into tissues and onto body surfaces such as the lining of the gut. Antibodies bind to viruses and virus-infected cells, helping to prevent spread of the invaders. In some instances, antibodies actually prevent viruses from infecting cells by blocking their receptor for entry and therefore are important in preventing later re-infection.
The relative importance of T and B cells in the control of virus infections is well illustrated by rare mutations that wipe out one or other lymphocyte type. Babies born with a mutation that eliminates their T cells die very rapidly of virus infections unless they live inside a germ-free bubble until they get a bone marrow transplant to correct the defect. Alternatively, babies with a mutation that prevents B cell development cope fairly well with virus infections but suffer from severe and persistent bacterial and fungal infections. However, they are generally protected from these infections during the first few months of life (as are healthy babies) by antibodies from their mother’s blood that cross the placenta in late pregnancy and are also present in breast milk.
The immune response to microbes is a complex but finely balanced operation, with the action of cells fighting the invaders counterbalanced by a group of cells called regulatory T cells. These produce cytokines that defuse a T cell’s killing mechanism and stop it dividing, so that once the microbe is defeated, the fighting cells die and the response is brought to an end, leaving only a skeleton crew of memory T and B cells ready for rapid action when the microbe appears again.
At the height of its activity, the immune response may be so pronounced that it actually does harm to the body. In fact, the typical, non-specific symptoms we experience with an acute dose of flu, such as fever, headache, enlarged tender glands, and general fatigue, are often not caused by the invading microbe itself but by the cytokines released by immune cells to fight it. On rare occasions, these immune-induced reactions may cause serious injury to internal organs, a result known as immunopathology. Examples include liver damage during infection with hepatitis viruses and the severe fatigue experienced by sufferers of glandular fever caused by Epstein-Barr virus. Alternatively, T cells or antibodies specific for viral proteins may, by chance, recognize, or cross-react with, a similar host protein. This can lead to damage to, or the death of, cells expressing the protein. This autoimmune process may be the basis of diseases such as diabetes, in which the insulin-producing beta cells in the pancreas are destroyed, and multiple sclerosis that results from destruction of cells in the central nervous system.
Some viruses have learned to play hide-and-seek with immune cells by protecting themselves from the ensuing onslaught and remaining in their host for long periods, even for life. Strategies employed by these viruses are as varied as they are ingenious, including evasion of immune recognition and/or obstruction of the immune response. Each step of the immune cascade, from the initial interferon release to the killer T cell attack and the later calming action of regulatory T cells, can be modified by one virus or another to promote their own survival.
For instance, HIV has several means of immune evasion including integration of its provirus into the host.