MediLexicon International -What Is Cancer? What Causes Cancer?

Carcinogenesis – how cancers start
As already indicated, cancer results when the changes required for the hallmarks of cancer have occurred. To understand how cancer develops, we now need to turn to how external factors bring about cancer – a process known as carcinogenesis. Fundamentally, cancer results from damage to DNA. All agents that damage DNA therefore are potential carcinogens – agents that cause cancer. The reverse is not true, however; not all agents that help cause cancer themselves directly damage DNA, though this always lies at the end of the process. Examples of cancer-causing substances that do not directly damage DNA include alcohol and the sex hormones involved in causing breast and prostate cancer. There are many sorts of carcinogens and many are well known – cigarette smoke and ionizing radiation, for example. Taking cigarette smoking, we know that typically it is necessary to smoke many cigarettes for many years for cancer to develop. This suggests that the process of carcinogenesis is slow and potentially has more than one step. From the discussion above, it would be predicted that mutations would be necessary in different sets of genes to cause the hallmark changes described by Hanahan and Weinberg. Such a chain of events was also postulated in the early 1990s and is now often referred to as the ‘Vogelstein cascade’, with each step in the chain representing a new mutation.
Dr Vogelstein’s group studied inherited bowel cancer, a condition in which there are a number of recognized pre-cancerous (also called pre-malignant) steps that could be identified in patients They collected tissues from patients and set about identifying which genes were abnormal in the various steps along the pathway from normal bowel lining to a clinically obvious cancer. It turned out to be possible to identify candidate genes that need to be damaged for each step of the cascade to occur. Subsequent work has demonstrated that similar cascades of events apply to all tumour types, though the individual genes involved and the sequence of damage vary.
One fruitful way to identify genes has been to study families with so-called ‘inherited’ cancers. The term is a bit of a misnomer as the cancer is not inherited in the same way as, say, a diamond necklace, that is, as an intact, fully formed object. What is inherited is a greatly increased risk of developing a disease early, often in a very florid, aggressive form. One such disease is called adenomatous polyposis coli (APC). Patients with the disease develop multiple benign adenomas from an early age. In time, some of these progress to cancer, and without treatment death typically occurs in the early 40s from bowel cancer. Studies of patients with the disease showed that they had abnormalities in a particular gene, which was named APC. The identification of the APC gene in these patients led to further study of the function of the gene, which turns out to function as an ‘off-switch’. If it is knocked out, an important check on cell growth is removed and adenomas form. As is often the way with inherited cancers, the much commoner, non-inherited cancers turned out to share similar abnormalities. Studies of non-inherited bowel cancer confirm that the APC gene is malfunctioning in around 80% of these sporadic cases, so the gene clearly has a key function in regulating the normal growth of the bowel lining.
Studies of inherited cancers thus often shed important light on the causation of the non-inherited counterpart disease. Study of these ‘cancer families’ helped identify key cancer-related genes such as APC, RB (linked to retinoblastoma, a rare childhood eye tumour), p53 (linked to Li-Fraumeni syndrome, in which patients develop multiple different cancers), and VHL (linked to von Hippel Lindau syndrome, a complex disorder that includes kidney cancer). In addition, examination of the varying natural history of the inherited disease helps us to understand what the normal function of these genes may be. All of the genes mentioned above are termed ‘tumour-suppressor’ genes, but this is a misnomer as this is not their primary role in the organism. As may be predicted from the APC gene, these genes are key regulators of the cell cycle (the first two aspects of the hallmarks mentioned above), and damage or deletion of function leads to uncontrolled growth. Examining the normal functions of these genes has shed important light on how the cell cycle is regulated. As lack of control of the cell cycle is a hallmark of cancer, many cancer treatments work by interfering with the cell cycle genes that are misfiring in the cancer cell. In addition, a new generation of cancer drugs, the targeted molecular therapies, is currently hitting the clinics and the news headlines.
These drugs work by targeting specific molecules known to be misfiring in the cancer. Not all inherited cancer genes are directly involved in the cell cycle, however. A good example is the VHL gene, originally identified in patients with von Hippel Lindau syndrome. Sufferers develop multiple abnormalities from an early age, including cysts in the nervous system, in particular the cerebellum (part of the brain involved in balance and coordination), spinal cord, and retina, together with kidney tumours both benign and malignant. The kidney tumours are typically bilateral, multiple, and occur from a young age. As for APC, the patient inherits one non-functioning gene; a single hit to the remaining gene leaves no functioning VHL protein in the cell. Given that renal tumours are relatively rare but are common in patients with VHL, this tells us that the chances of a given gene suffering one hit are relatively high, but suffering two hits takes much longer, hence sporadic tumours are single and have a much later age of onset.
Detailed study of the VHL gene has revealed that it is involved in sensing the oxygen levels in the cell. If oxygen is low, this leads to the production of signals to surrounding cells to start growing new blood vessels. In other words, it regulates angiogenesis, a key hallmark of cancer.
Further studies have shown that these changes are sufficient to drive the cancer cell in the test tube, and the replacement of the VHL gene in these models will reverse the cancerous characteristics of the cells. Furthermore, the kidney tumour type found in VHL patients, called renal cell carcinoma, is characteristically very rich in blood vessels, as may be predicted from the gene function. Study of sporadic (non-inherited) renal cell carcinomas has revealed that VHL is mutated in around 70% of cases, making the VHL/angiogenesis pathway an attractive target for therapy. Research into new VHL-based treatments for kidney cancer, a notoriously difficult cancer to treat once it has spread, has proved very fruitful, with six agents licensed since 2006 and several more pending for a disease for which only two agents had been licensed in the previous 25 years. All of these agents target aspects of the pathway identified by the genetic research summarized above.
Non-inherited cancer
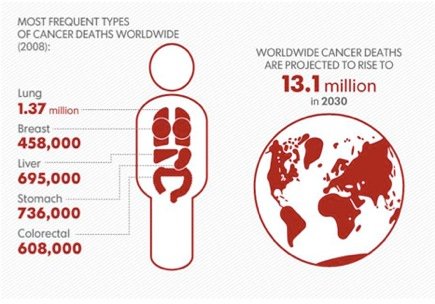
While inherited cancers shed important light on the classes of genes involved in cancer, the majority of cases of cancer do not result from an obvious inherited predisposition. As we have seen in the major causes of cancer death worldwide arise from tumours in the lung, stomach, liver, colon, and breast. Of these cancers, lung cancer is strongly linked to cigarette smoking, and liver cancer to infection with the hepatitis B virus, with a significant role for alcohol consumption. Cancers of the digestive tract are presumed to be linked to diet, but the precise causation is still poorly understood. Likewise, breast cancer (and prostate cancer in men) is clearly linked to both dietary and hormonal factors. How may these diverse influences act to produce the changes required to generate a cancer described above?
Lung cancer is the best understood example of how a carcinogen in the environment can interact to generate a cancer. The risk is clearly linked to amount of tobacco consumed – there is a dose effect – and the duration of consumption. Smokers who give up tobacco before getting cancer have a decreasing risk of developing the disease after stopping. In terms of a model like the Vogelstein cascade, smoking must be responsible for inducing the first steps of the cascade and continued smoking must also induce the subsequent steps. In older models of carcinogenesis, the initial step was often referred to as initiation and the subsequent steps as promotion of tumour growth, with a final step termed transformation. These terms still have value and in the laboratory, agents that convert non-malignant cell growths into cancerous ones are often referred to as transforming the cells.
Analysis of tobacco smoke has revealed a host of agents that will result in transformation in cell culture systems. Detailed study of these smoke constituents has revealed the precise molecular mechanisms at work, down to the mode of interaction with the DNA double helix. One of the key culprits is called benzopyrene, and careful research has demonstrated it will bind to the DNA helix, damaging the structure. As mentioned above, there is clearly a need for DNA damage – an initiating event followed by a generally prolonged period of further damage accumulation, sometimes referred to as promotion – before a final transforming event turns the pre-cancerous lesion into a full-blown cancer. In the case of tobacco, the process appears to be driven by continuous exposure to tobacco smoke, which has direct DNA-damaging properties. For other diseases, in particular breast and prostate cancers, the role of promoter is taken by the individual’s own hormones. As indicated, risk of breast cancer is influenced by duration of exposure of the breast to cyclical female hormones – hence early menarche and fewer pregnancies with no breast-feeding results in increased risk. The inference of this is that the continued cycles of changes in the breast induced by the menstrual cycle magnify any initial DNA damage done by some form of environmental carcinogen. A similar effect is seen in prostate cancer, in that men castrated early in life (for example, eunuchs) have a very low risk of prostate cancer compared to their peers who presumably are exposed to the same environmental carcinogens. A similar role is played by alcohol in liver disease. Alcohol, as already noted, is not a direct carcinogen – it will not damage DNA. However, heavy long-term abuse of alcohol induces cycles of damage and repair in the liver, with increased cell turnover. As with the cyclical changes in the breast, this continual increased activity serves to magnify the harm done by the DNA-damaging agents which must also be present, increasing the opportunity for accumulation of further DNA damage and the development of cancer.
In the case of liver cancer, we also have detailed knowledge of the most frequent carcinogen – hepatitis B infection. The disease is a massive cause of suffering worldwide, but particularly in China and other parts of Asia where up to 10% of the population is chronically infected. There are lower rates in India and the Middle East, with rates less than 1% in Europe and North America. The risk of developing chronic infection is highest in those infected in infancy. Since 1982, a vaccine to HBV has been available. With vaccine programmes in place in various countries for some years, this has allowed scientists to complete the final test of the link between a virus and cancer – if the link were causal, preventing infection should and did prevent the disease. The precise molecular mechanism whereby the virus causes cancer is still being studied, but, as with smoking, the evidence for causation is now compelling.
Moving on to another cancer linked to infection – cervical cancer – we can see a similar story emerging. It was observed in the 1920s that cervical cancer was more common in women who had had high numbers of sexual partners, in particular prostitutes, and it was rare in nuns (except for those who had been previously sexually active), suggesting an infective, sexually transmitted cause. The disease was shown to be linked to infection with the human papillomavirus (HPV) by Harald Zur Hausen in 1976. Dr Zur Hausen found HPV DNA in both genital warts and cervical cancer. He received the Nobel Prize for Medicine for this discovery and his subsequent work in the field, which demonstrated the precise molecular links between the virus and the cancer. The virus produces various proteins which interact with two genes called Rb and p53, both key controllers of the cell cycle, providing an obvious route for generating a cancer.
The development of a vaccine against HPV and hence cervical cancer has proved more technically challenging than the HBV vaccine. However, the linkage between chronic viral infection and cancer allowed the study of the pre-malignant stages of cervical cancer. This led to the discovery that these could be identified in smears of cells taken with a wooden spatula from the cervix and then examined under the microscope. Identification of the pre-cancer stage, called cervical intra-epithelial neoplasia (CIN) or carcinoma-in-situ (CIS), allowed preventative treatment. Most European and North American countries have comprehensive screening based on the cervical smear test. These programmes have been estimated to have saved many thousands of lives. More recently, a vaccine against the varieties of HPV linked to cervical cancer became available in 2006 and is beginning to appear in public health vaccine programmes for girls as a way of preventing infection, with consequent reduction in cancer risk. There is some controversy about the vaccine as some interpret it as a way of protecting against the risks of promiscuity. However, protection against one sexually transmitted disease does not lower risks from others such as HIV. In addition, the vaccine will protect women against the risks of prior promiscuity in their partners, something they have no control over whatsoever. It will, however, take 10 to 20 years for this benefit to be seen, as this is the typical time lag between HPV infection and the development of cancer.
How is cancer treated?
Cancer treatment is complex and typically will involve input from a number of different groups, ranging from a wide assortment of doctors, including general practitioners (family doctors), surgeons, oncologists, pathologists, radiologists, palliative care specialists, as well as a huge number of other trained personnel – nurses, radiographers, physiotherapists, technicians in laboratories and radiotherapy departments, theatre orderlies, the list goes on and on. The details of organization for these different groups vary enormously from country to country and are a function of both the politics and economics of healthcare.
To try and get around this problem, I will present the organization of cancer treatment as a journey from symptoms to diagnosis, treatment, follow-up, and palliative care for those experiencing incurable recurrence. Different healthcare systems will process these events in a variety of ways, but by and large, the underlying principles are pretty universal. The final part of the chapter gives an overview of the main different classes of treatment, such as surgery, chemotherapy, and radiotherapy.
Initial diagnosis and investigations
Most patients still present with symptoms such as a persistent cough or problems such as blood appearing in the urine. Significant numbers are also picked up by screening programmes, either organized on a systematic basis (for example, for breast and cervical cancer) or more informally (such as PSA testing for prostate cancer). Some cases are picked up as incidental findings in the course of investigation for some other problem. For example, an abdominal scan may detect an asymptomatic tumour in a kidney. I will return to these groups of patients later.
Most patients will present to their doctor with some sort of symptom they have noticed and which they are worried about. Although symptoms, like people, come in an unlimited number of varieties, they can mostly be grouped into those causing disruption of normal function, such as a brain tumour disrupting normal movement, or abnormal symptoms due to damage by the tumour, such as bleeding, pain, or cough. The period between initial symptoms and diagnosis of cancer may be very short or may sometimes run to years. Sometimes the delay in diagnosis is down to misinterpretation of symptoms by health professionals, sometimes deliberate self-neglect or self-deception by patients, and sometimes a mixture of the two.
Unsurprisingly, the perception that an opportunity to make an early diagnosis has been missed can cause severe subsequent problems in the relationship between the patient and their doctor, often at a time when they need them most. Family doctors have a tough time in this respect. For example, headache and backache are common symptoms, and in the vast majority of cases have benign causes that may need symptomatic remedies but do not need extensive investigation. Occasionally, of course, these symptoms may indicate an underlying brain or spinal tumour. Another example is the presence of blood in the bowel motions. All medical students know that this may indicate bowel cancer. All family doctors will know that for patients in the ‘at risk’ age range for bowel cancer, the presence of conditions like haemorrhoids (an irritating condition of the lower anus that can bleed) are virtually universal. How do they then set about distinguishing the banal from the severe (but rare) without grossly over-investigating their patients? The answer often lies in another basic skill taught in medical school – the art of taking an accurate history. Thus, a sudden and unexpected change such as severe bleeding mixed in with motions is much more likely to be due to a cancer than a small quantity of bright red blood being seen smeared on the toilet paper occurring over a period of years.
Screening for cancer
In an ideal world, we would be able to offer tests that picked up cancer before it reached the more serious stages, allowing early intervention and a much greater prospect of cure. Such a process is called screening and is now available for a number of cancers: breast, uterine, cervix, and bowel. In addition, the PSA blood test is a potential screening test for prostate cancer, but its use remains controversial. It is helpful to describe the characteristics of an ideal screening test and then examine how these tests shape up in practice.
This is illustrated in the following example:
Features of an ideal screening test (Source: WHO)
• The target disease should be a common form of cancer, with high associated death rate
• Effective treatment, capable of reducing the risk of death if applied early enough, should be available
• Test procedures should be acceptable, safe, and relatively inexpensive In addition, we need to consider:
• True positive rates: Sick people correctly diagnosed as sick
• False positive rates: Healthy people wrongly identified as sick
• True negative rates: Healthy people correctly identified as healthy
• False negative rates: Sick people wrongly identified as healthy
Relation between results of liver scan and correct diagnosis
Thus the sensitivity (Patients with liver disease and abnormal scans/all patients with a positive scan) = 231/231+31 = 0.88 and the specificity (Patients with normal scans and no disease/all patients with normal scans) =54/(27+54) = 0.67
A further measure is the positive predictive value (the proportion of patients with abnormal scans who have liver disease) = 231/(231+27) = 0.89 and the negative predictive value of a negative scan (the proportion of patients with normal scans who have no liver disease) = 54/(32+86) = 0.45
For a test in the clinic, this is pretty good – a positive scan in someone suspected of having liver disease is a pretty good indicator that the person has the disease. How does this fare as a screening test, then?
To illustrate the difference between using a test for diagnosis in someone already known to be ill and screening for disease in people with no symptoms, we can look at the figures for breast cancer. Let us suppose that the rate of missed cases (the specificity) in those who we test is 10% and that the level of early, undetected disease is 1 in 500 people. If we now test 100,000 subjects, an ideal test would yield 200 positive tests in the cancer sufferers and 999,800 negative tests in those without the disease. However, our test, though good, is not perfect and will only detect 180 of the 200 cases, leaving 20 people wrongly reassured. Conversely, the test is also not completely specific. Let us say that 95% of those without the disease will test negative but 5% will wrongly test positive. When we apply this to our screening population, we see that this means that 5% of the 99,800 without the disease will falsely test positive. This works out as 4,999 false positive tests in people without the disease. This means that only a minority (180/4,999 = 4%) of those with a positive test actually have the disease, but 4,999 – 180 = 4,819 people have had a nasty scare. Furthermore, 20 have been falsely reassured and will go on to present with cancer anyway, possibly detected late as they may ignore the symptoms, believing themselves to be cancer-free. However, the overwhelming majority of those with a negative test (99,800 – more than 99%) really were free of the disease, so a negative test is pretty reassuring.
These worked examples are important, as they illustrate the limitations of screening tests which at first sight sound pretty good. In point of fact, the figures above are the best figures available – sensitivity and specificity fall in younger women (probably because their breast tissue is denser, making it harder to see abnormal lumps), leading to more incorrectly categorized cases. Furthermore, while the cost of the test itself is small, the cost of chasing up the false positives is much larger and needs to be factored into the costs of the screening programme.
There is a further problem when working out the benefit of screening. In our example above, we will identify cases of cancer earlier than would have happened without screening, potentially improving treatment prospects. However, with breast cancer, the cure rates are good, with three-quarters of diagnosed women being long-term survivors. This leaves the quarter who are destined to do badly, who are the main potential beneficiaries of screening. This is a relatively small number in relation to the numbers of tests carried out, and the downside is over-investigation of healthy women without breast cancer.
Investigating suspected cancer
Whether the patient has been picked up by a screening programme or has presented to their doctor with worrisome symptoms, the next step is to carry out further tests to confirm or exclude the diagnosis. Diagnosis is usually based on a tissue sample (biopsy) of the affected organ, preceded by clinical examination by a doctor, imaging, and blood tests. Ideally, cancer would be investigated using non-invasive imaging tests. In practice, in almost all cases the diagnosis needs to be confirmed by examining a tissue sample in the laboratory. Imaging is key to deciding where and how to obtain tissue. Modern cross-sectional imaging either with X-rays – computed tomography, or CT, scans – or using magnetic resonance imaging (MRI) can give remarkably detailed pictures of internal organs and suspected tumours. However, even the best imaging is unable to show with certainty whether a mass is cancerous and also, even if the diagnosis of cancer is highly likely, exactly what sort of cancer. Occasionally the imaging is sufficient. For example, an elderly, frail, life-long heavy smoker with suspected lung cancer on a chest X-ray and who is unfit for any treatment may be spared the discomfort of a confirming biopsy. One or two other scenarios may also not require a biopsy – patients with extensive cancer deposits in bone (a common site of spread for prostate cancer) on imaging and a grossly elevated serum prostate specific antigen (PSA) can be reliably diagnosed as having widespread prostate cancer with no biopsy. In all of these cases, the abnormalities are evident. However, even for radiologically obvious lesions such as these, a biopsy is generally required to determine the exact cancer type and hence the appropriate treatment.
The role of the pathologist in cancer diagnosis
The pathologist assesses small tissue samples taken, for example, via a needle – biopsies. Occasionally, for example in renal cancer, the initial material may be from a surgically removed organ, such as the diseased kidney. Mostly, this is done by mounting very thin slices of the removed tumour on slides and then carrying out a range of stains which highlight particular features of interest. The stained slides are then examined by the pathologist using a microscope. A commonly used stain is haematoxylin and eosin (usually called H&E) which highlights the various components of the cell such as the nucleus. Increasingly specialized stains are used which help further characterize the tumour. An example would be staining for the oestrogen receptor in breast cancer which helps predict the response of the cancer to both chemotherapy and hormone therapy. There are a rapidly growing number of available tests, mostly based on monoclonal antibodies (which are also increasing rapidly as a form of treatment). In addition, tests can also be done to look for changes in the expression of particular genes or to look for the presence of particular mutations or rearrangements in chromosomes.
The primary question for the pathologist is: ‘is it cancer?’ If the answer is ‘yes’, then secondary questions include the specific type – in other words, in which organ did it start and which subtype. In addition, cancers are graded in terms of aggressiveness, typically on a scale of one (low) to three (high). Some cancers, for example prostate cancer, lymphoma (cancer of the lymph glands), and sarcomas (cancers of the connecting and structural tissues such as bone, muscle, or cartilage), have different grading systems, but the same principles apply. All of these systems are based on the size and shape of the cancer cells and how they compare to the normal cells in the organ in which they originated.
More recently, and increasingly, additional sub classification is based on molecular markers present on the cancer. These can be defined as characteristic features based on excessive levels of particular markers either in the tumour itself or circulating in blood (or sometimes present in urine). Probably the best-known example of a molecular marker is HER2 in breast cancer. This was initially described as a marker of poor outcome in breast and ovarian cancer by Dr Denis Slamon from UCLA in the late 1980s. This led to the development of the drug trastuzumab (Herceptin), intended to target cells with excessive amounts of the protein (termed over-expression). Landmark trials, initially in patients with advanced disease and subsequently in newly diagnosed patients, showed that the drug significantly improved outcomes for the 25% of women with tumours with high levels of the HER2 protein. Staining tumour samples for HER2 expression thus gives important information about prognosis (treatment outcomes) and also helps guide the choice of treatment.
The other major role for the pathologist in cancer care is the assessment of specimens resulting from surgical removal of organs containing cancer. In addition to the questions posed above, which will be reassessed with the larger specimen, the pathologist is also addressing issues such as:
• Is the tumour confined to the organ that has been removed at surgery?
• Are the surgical resection margins (the edges of the specimen) free of tumour?
• Is there spread to other associated structures such as lymph glands?