VIDEO
Purchase Weight Loss drugs, Buy Acomplia cheap generic Shop
Acomplia
ACOMPLIA IS THE trade name of rimonabant (generic name), which has been approved in the European Union (EU),
As of March 2007, rimonabant (the proposed
Rimonabant is the first inverse agonist discovered for the cannabinoid-1 receptor (CB1R). Although rimonabant binds to the CB1R, it is not a cannabinoidcannabinoid-like compound and bears no structural resemblance to cannabinoids.
In cell-based in vitro assays, rimonabant inhibits the intrinsic activity of CB1R and produces an effect opposite that of an agonist; hence, rimonabant is classified as an inverse agonist. It also inhibits the binding of cannabinoid agonists and endocannabinoids to the CB1R, so it is sometimes referred to as an antagonist.
In experimental animal studies, rimonabant does not exhibit cannabimimetic properties and it inhibits many agonist-induced effects. In rodent studies, rimonabant can cause significant weight loss or reduction of weight gain.
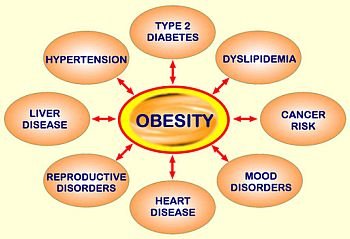
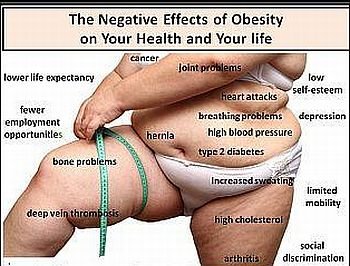
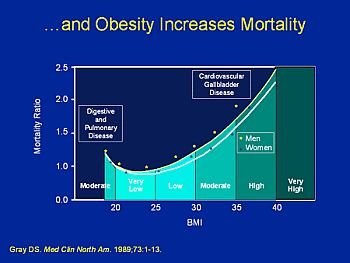
In clinical trials involving obese patients, rimonabant has been shown to cause weight loss and reduction of waist size. Beneficial secondary end points have also been demonstrated, including increased high-density lipoprotein cholesterol (HDLC) and improved glycemic control.
Rimonabant at 20 mg is generally well tolerated. As described in the Summary of Product Characteristics approved by the EU regulatory agency, the most common adverse reactions resulting in discontinuation include nausea, mood alteration with depressive symptoms, anxiety, and dizziness.
See also: Cannabinoid System; G-Protein Coupled Receptor.
Bibliography.
C. Curioni and C. Andre, “Rimonabant for Overweight or Obesity,” Cochrane Database of Systematic Reviews (v.2006/4, 2006); Zimulti Acomplia Report, www.acompliareport.com/ (accessed April 18, 2013).