Migraines Caused By Bipolar and Depression Disorders
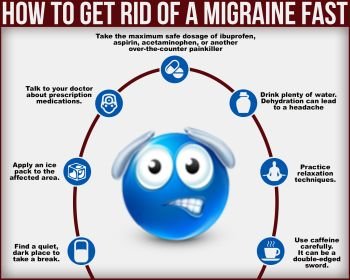
18 Signs You’re Having a Migraine
Research and the future
You need to understand what scientific research is about before you can understand how to interpret the results that make the headlines. You need to be sure that the information presented is not just spin before you can decide that a treatment or intervention is worth the risk and does actually offer any benefit. I hope that what follows answers some of your questions as to what research is all about and what the future might offer the headache sufferer.
The internet has ensured that a vast amount of information is readily accessible. However, you need to be sure that the quality of that information is high and relevant to you. The best place to start is usually the UK-based headache charity websites. They will often have links to other reputable and well-researched websites. Try to stick to UK-based sites, as some drugs and interventions mentioned on sites in other countries may well not be available in the UK.
RESEARCH: THE WHYS AND WHEREFORES
Why is research important?
There would be no new drugs without research. Research is crucial to our understanding of how the brain works – without that knowledge, scientists and clinicians would not be able to suggest ways of tackling medical conditions and altering or modifying the progress of disease.
Without research we would not have brain scanners, we would not have triptans and we would not know the best and safest ways of repairing holes in the heart or removing brain tumours.
Why is research relevant in migraine?
Research is relevant because without asking questions we cannot understand what is happening, and without knowing what is happening we cannot find ways of changing or modifying it.
In the context of migraine, if scientists had not found out about 5-HT receptors and what they do, we would not have had triptans.
As more is understood about 5-HT receptors, better triptans or other drugs can be developed that work more effectively.
What is ‘anecdotal’ evidence, and why do we have to prove that something works when people I know tell me it works?
When choosing a holiday or a place to stay, listening to someone else’s experience can be helpful in deciding whether you want to go to a particular place or stay in the same hotel. The downside is that if, when you get there, the weather is bad or the hotel is run by different people, your experience might not be as good – so you would never go there again.
A few days, weeks or months later, someone else goes there and has a great time – do you go back or not?
That is decision-making based on ‘anecdotal’ evidence. If you reflect on the example above and transfer that sort of decision-making across to medicine, the issues raised and challenges faced become potentially quite difficult and in some instances even life-threatening.
Any decision you make needs to be sound, safe and cause no harm, hence the need for research and, above all, good research.
What is good research?
Good research is about making sure you ask the right question, in the right way, and set about answering that question using all the right resources, in the right way.
In the context of migraine and triptans you need to evaluate a series of features and factors.
To name just a few:
• Is this drug safe?
– in everyone?
– in the young?
– in the elderly?
– in those who are pregnant?
• Does the drug work?
• Is there a right dose or an optimal dose?
– does the maximum dose cause too many side effects?
– does the dose vary in different age groups?
• Is the patient headache-free?
– in one hour?
– in two hours?
– in four hours?
• Is the headache only eased?
Any piece of research must be able to answer all of these questions and many, many more.
How should good trials be constructed?
Agood trial needs a clear starting point, which means asking a clear and precise question or series of questions.
In the context of preventative drugs or interventions:
• Will X reduce the total number of headache days?
• Will X reduce the total number of headache days by more than 50%?
• Will X make the headache easier to treat with current acute treatment?
• Does X cause side effects?
• What are those side effects?
• Are those side effects sufficient to cause the patient to stop taking the drug?
• Does X perform better than a placebo?
• Does X perform better than the drug the patient was using before?
• Does X perform better than the ‘gold standard’ or most commonly used drug for, in this case, migraine prevention?
The patients recruited to the study must have the correct diagnosis; in this case the standard is migraine as defined by the International
Headache Society
There must be enough people recruited to the study to ensure that any statistical analysis will have value. There should at the very least be a placebo or ‘dummy’ group, and, if feasible, a comparison with a drug already used to treat the condition. No one, that is to say neither the people who are co-ordinating the study and reviewing the patients in the study nor those involved or participating in the study, should know if they are taking active drug or placebo (this is called ‘double blind’). If there is a placebo group, it is always interesting to swap this group over to the active drug and assess their response, and vice versa (referred to as ‘cross over’).
There has to be a baseline period to collect and collate information, in the form of diary cards, to better understand the group being studied. Patients in the trial must stop all other drugs that may have been used, usually for a minimum of four weeks.
There needs to be a minimum number of headache days and a maximum number of headache days recorded within this baseline period, so that patients meet clear recruitment criteria.
Once the trial drug is started, there must be a clear method of increasing the dose, over fixed time intervals, to monitor how effective it is and any side effects (adverse events). There is often a balance between effectiveness and side effects. There must also be a clear way of recording side effects, and identifying at what dose these develop.
The study drug has to be taken long enough to ensure that the full effect of treatment (e.g. maximal reduction in headache days) is recognised and seen, and a follow-up to assess how long the benefit is felt (e.g. for three months? six months?) or sustained.
How is a decision made, based on trial information?
This is never easy but initially it is important that enough patients were recruited to give value to the study: 300 are better than 30.
The statistics are examined to see whether they reach ‘significance’ and that should be highlighted in the conclusions or even the initial abstract of the report. It will often say that the ‘difference was statistically significant’ and quote a ‘probability’ (p) value – the result of the statistical calculation made.
There may be times when the study did not go on long enough and so the results did not reach statistical significance, or the study recruited enough patients to reach statistical significance in one area but not in another.
It is often difficult to tease out the detail from an article or scientific paper but the bottom line in acute drug treatment for migraine is:
• Is the patient headache-free at 2 hours?
• Is there a sustained pain-free response?
• Does ‘rescue’ medication have to be taken?
• Are the side effects less than with a placebo?
And, for preventative drugs:
• Is there a reduction in the total number of headache days?
• Is there a greater than 50% reduction in headache days?
• How long did the drug have to be taken for an effect to be seen?
• At what dose did the drug need to be taken for maximum benefit to be achieved?
• Are the side effects less than with a placebo?
As is often the case, there is a trade-off between effectiveness and side effects. Decision-making is never easy, and rarely straightforward.
There is no drug out there that will fix all of your headaches all of the time. Hopefully, as more is known and understood about headache symptoms, newer drugs will be developed that work well.
How can I become involved in research?
There are always trials going on somewhere, and the best source of information is probably the headache charities.
Alternatively, many of the specialist headache centres around the UK will be involved in research of some sort, although not always drug-based research. Some research uses special types of brain scan to understand more about what happens in the brain; other trials are about understanding more about what effect migraine and other headaches have on your life – impact studies. Other types of research are looking at interventions such as acupuncture or osteopathy to see if they help headache patients.
How are patients selected for trials?
Patients tend to be selected on the basis of their headache diagnosis in the first instance, then on their age and whether they are interested in being involved. You need to be aware that you may not be given any active drug or intervention but will be given the dummy or placebo drug or intervention instead.
There are always strict inclusion and exclusion criteria that depend on the question being asked. If the trial is for acute treatments, you have to be prepared to use only the trial drug for a series of attacks; often patients are selected only if they have never treated their
migraine with anything other than simple painkillers; it just depends. If the trial is for a preventative drug, there are other inclusion and exclusion criteria that apply. For example, the number of headache days each week or month, how many different preventative drugs you have tried in the past and at what doses you tried them, whether you took them for long enough.
How do I know that a new drug is safe to take?
This is always difficult to answer, as newspaper headlines sometimes attest. Drugs will have gone through significant assessment and evaluation before they are given to humans, but all animal and computer models are just that – a model. There is no way to predict exactly what effect a drug will have on the human body until it is administered for the first time.
If you want to become involved in a drug trial, you will be given a lot of information to read, absorb and understand. It is your responsibility to make sure that you clearly do understand and ask all and any questions that need an answer before you are ready to sign the consent form.
In many ways there is no way of knowing a drug is safe until it has been used for many years. Drugs that have been used for decades have been found to be associated with previously unrecognised risks.
Sometimes it is about weighing up the balance of potential and theoretical risk against the benefit you get.
If I agree to be involved in a trial and I feel that I am getting worse, can I opt out?
You can opt out at any time. When you do so you will be asked a series of questions so that the researchers doing the trial can understand why: this helps them assess all potential problems associated with the drug or intervention they are investigating.