Understanding Targeted Therapies for Cancer
Targeted Therapies
A seventy-five-year-old woman with non-Hodgkin’s lymphoma was experiencing weakness and discomfort from enlarged lymph nodes in her neck. She had numerous other medical problems, and her health was too poor for chemotherapy. She received a targeted therapy called Rituxan for one month. She experienced only mild fatigue from the medicine. Her lymphoma responded completely, and she remains without evidence of the disease six years later.
A sixty-five-year-old man developed a recurrence of kidney cancer twenty years after it was removed. The cancer was extensive in his liver and pancreas, and he was extremely weak; it did not respond to interferon, and chemotherapy was felt to be useless. He was treated with a targeted pill called sunitinib (Sutent). Almost overnight he felt better, and after two months his cancer had shrunk dramatically in size. The cancer was controlled in this way for two years, with minimal side effects from treatment.
A fifty-year-old woman was admitted to our hospital with fever, abdominal pain, and jaundice. Her health had been declining for several months; now she could barely walk and couldn’t eat. She was found to have colon cancer that had formed extensive metastases throughout the liver. We initiated treatment with three chemotherapy drugs (known as FOLFOX) plus a targeted therapy called bevacizumab (Avastin). After a week she could leave the hospital. After two months of regular treatments, she was pain-free, gaining needed weight, and back to full activity levels. CT scans showed that the liver tumors had shrunk by more than 50 percent. After six months of treatment the liver tumors were barely detectable.
TARGETING THE CANCER GENERATOR
The best way to cure a medical disorder is to understand its causes and then block or reverse them. For example, coronary artery disease can be caused by too much cholesterol (along with other risk factors); one way to lower cholesterol levels is with “statin” drugs, which block an enzyme critical for its synthesis in the liver. Ulcers are caused, in part, by excess stomach acid; drugs that block the molecules responsible for acid production, such as omeprazole (Prilosec), help heal ulcers. The targeted therapy of many human ailments has proven to be effective and safe. A similar approach to the treatment of cancer has been the light at the end of the tunnel of cancer researchers for the past fifty years.
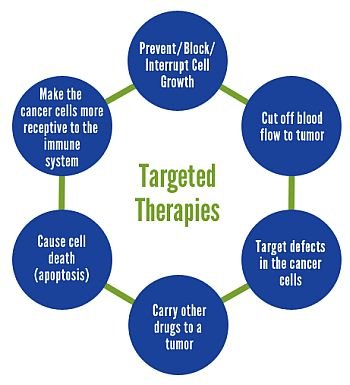
Some genes are overactive, leading to excessive cell growth, whereas others are underactive, allowing this growth to be unrestrained. Thus most targeted cancer therapies aim to shut down the function of an overactive gene or replace an underactive one (this is much more difficult). Many of the overactive genes are part of the communications network I described above; they give rise to receptors and signaling molecules that are always turned on. Like an electrical circuit that cannot be shut off, overactive receptors and signaling molecules repetitively fire, creating supercharged cancer cells that have their own internal generators.
The drugs described below aim to stop these misfiring: the result may be the restoration of calm and the slowing of growth or, better yet, the death of cancer cells. There are thousands of potential targets in cancer cells; advances in chemistry and computing are greatly accelerating the discovery of drugs that can bind too many of them. This class of drugs will likely represent the great bulk of new cancer therapies in coming years.
TARGETING RECEPTORS
Drugs like cetuximab (Erbitux) for colorectal and head and neck cancers, rituximab (Rituxan) for lymphoma, and trastuzumab (Herceptin) for breast cancer are administered intravenously. Once in the bloodstream, they act like heat-seeking missiles, locating cancer cells wherever they lurk and gripping onto them via one specific receptor target (among thousands of receptors) that projects from the outer surface of the cells. The result is that the receptors stop transmitting growth signals inside cancer cells.
Many drugs in this class are modifications of large molecules called antibodies, which our bodies normally generate against infectious agents such as bacteria. Cancer-fighting antibodies are also called monoclonal antibodies. Because of their structural similarity to infection-fighting molecules, cancer-fighting antibodies are also believed to stimulate the immune system to attack cancer cells once they attach to them. Monoclonal antibodies coat the surface of cancer cells, shut down receptor activity, and (hopefully) lead to cancer cell death. A second way to target cancer receptors is with drugs that pass inside the cell and bind to the parts of receptors in direct communication with the inner world of cells. Although the receptor can still receive signals from the outside, it cannot transmit these signals internally if it is bound by these drugs. The drugs in this class are commonly called small molecule inhibitors because they can pass freely into cells (their small size also allows them to be given as pills). Examples include erlotinib (Tarceva), approved for lung and pancreatic cancer; sunitinib (Sutent), approved for kidney cancer and GIST tumors; and lapatinib (Tykerb), approved to treat breast cancer.
TARGETING SIGNALING MOLECULES
The communications pathway from surface receptors to DNA involves hundreds of different molecules. Silencing a critical signaling molecule in this network can disrupt the growth and survival signals on which cancer cells rely. Without these signals, cancer cells can die. By analogy, suppose a courier has to deliver a package on the other side of a river and has to cross a bridge. But when he arrives at the bridge, he finds that it has collapsed; the package will not reach its destination. By shutting down important targets that a growth signal must use to travel inside a cell, drugs called signal transduction inhibitors effectively “collapse bridges” throughout cancer cells. Examples of signal transduction inhibitors include imatinib (Gleevec) for CML and GIST tumors and sorafenib (Nexavar) for kidney and liver cancer. Many new drugs in this class of cancer fighters will become available in the future.
TARGETING BLOOD VESSEL GROWTH: ANGIOGENESIS INHIBITORS
For a cancer to survive, it needs oxygen and nutrients delivered by an adequate blood supply. A growing cancer stimulates certain cells in nearby blood vessels to make new blood vessels for itself. These are called endothelial cells; the process of making new blood vessels is called angiogenesis.
Angiogenesis inhibitors are drugs that stop endothelial cells from forming new blood vessels. They do this by jamming their communications networks. Just as in cancer cells, endothelial cell growth signals are transmitted through receptors and signaling molecules. Drugs such as bevacizumab (Avastin), sunitinib (Sutent), and sorafenib (Nexavar) affect the function of these molecules, causing blood vessels that feed a cancer to wither and die. Angiogenesis inhibitors may also have a direct effect against cancer cells because some of their targets are also present on cancer cells themselves, not just on their surrounding blood supply; they may thus have a dual cancer-fighting effect.
TARGETED THERAPIES
Are targeted therapies “magic bullets”?
Although targeted therapies were developed with the hope that they would be magic bullets that would neatly eradicate cancer through the selective targeting of one critical molecule, in general they have fallen short of this lofty goal. No cancer is considered curable by treatment through a targeted therapy alone, except perhaps for some cases of CML treated with Gleevec. This does not mean that targeted therapies have not helped many cancer patients, because they have. It probably means that the bar was set too high and that researchers expected targeted therapies to be “home runs” instead of the singles and doubles they have turned out to be.
The reason for the muted success of targeted therapies is that most cancers are caused not by one genetic derangement but by several; no one target functions as an Achilles heel. The next generation of targeted therapies is being designed to block several communication molecules from functioning. In this way the “magic bullet” concept will likely give way to the “magic shotgun.”
Can targeted therapies be used instead of chemotherapy?
Targeted therapies have not eliminated the need for chemotherapy. The three patient vignettes presented at the beginning of this section illustrate the three uses of targeted therapies: (1) as an alternative to chemotherapy when both have activity against a cancer (such as in low-grade lymphomas, when Rituximab alone may be an effective therapy); (2) where chemotherapy has a limited role (such as in kidney cancer and GIST tumors); and (3) to complement chemotherapy (such as in breast, lung, and colorectal cancers). The combination of the most effective chemotherapy drug(s) plus a targeted therapy is becoming a prevalent way to treat cancer.
For example, because all cancers need a blood supply to grow, angiogenesis inhibitors are being successfully combined with chemotherapy to treat a broad array of cancers. Because not all targeted therapies work well with all types of chemotherapies, clinical trials are necessary to establish which combinations work best.
If angiogenesis inhibitor drugs decrease blood flow to a tumor, how can they improve the effectiveness of chemotherapy?
For any cancer-fighting drug to work, it must have access to the cancer through the bloodstream. It is therefore counterintuitive that angiogenesis inhibitors, which diminish tumor blood flow, should improve the effectiveness of chemotherapy. Here is one possible explanation why it works: Consider a new ball of string, neatly wound around and around into an organized pattern. Next, imagine giving that ball of string to a playful cat and returning an hour later. You would find a disorganized heap of string, with the strands nicked and torn in many places. Now imagine that the string represents blood vessels: the new ball represents the pattern of blood flow in a normal organ, whereas the string damaged by the cat represents the course of blood vessels in a tumor.
Angiogenesis inhibitors actually help reorganize the frantic pattern of tumor blood vessels by repairing the nicks that make them leaky and diverting blood from the most injured ones. As a result, tumor blood vessels become “normalized,” and blood distribution actually improves, though it is diminished overall. The result is that chemotherapy has improved access to cancers when given with angiogenesis inhibitors.
Do targeted therapies cause side effects?
Like any drug taken for any purpose, unintended effects may occur with these medications. Generally speaking, targeted therapies are easier to tolerate-less hair loss, smaller declines in blood counts, and less nausea. For example, Rituxan is much gentler than chemotherapy for lymphoma, and Gleevec causes far fewer side effects than interferon or a stem-cell transplant for CML.
Still, substantial side effects may occur with some targeted therapies, and they tend to increase the toxicities of chemotherapy when used in combination. For example, drugs that block a molecule called the epidermal growth factor receptor (EGFR) on the surface of cancer cells, such as Erbitux, Vectibix, and Tarceva (called “EGFR inhibitors”), often cause an acne like rash on the face and upper body that may be severe; helpful medicines include topical hydrocortisone cream 1 percent or 2.5percent, clindamycin 1 percent gel, pimecrolimus cream (Elidel), and oral doxycycline or minocycline twice daily. In the case of severe skin reactions, the targeted medicines may need to be stopped temporarily or their dosages reduced to allow the skin to recover. Interestingly, some rash is a good thing: patients who develop any rash with EGFR inhibitors usually experience better control of their cancer than those who do not. It is important to inform your oncologist when a faint rash begins and not wait until it has become very bothersome and prominent; early intervention is the best way to prevent severe skin reactions.
Another type of skin reaction to cancer-fighting medications, called “hand-foot syndrome,” may occur with the targeted therapies Sutent and Nexavar as well as with some chemotherapy drugs, such as Xeloda, Doxil, and 5-FU. In hand-foot syndrome, the hands and feet become dry, reddened, painful, and swollen; there may also be skin breakdown and blisters on the palms and soles. Moisturizers and emollient preparations such as Bag Balm and Udder Cream are often helpful, and your oncology team may recommend other measures. Again, early intervention is the key to preventing severe hand-foot syndrome. Finally, angiogenesis inhibitors constrict blood vessels not only inside tumors but also in other parts of the body. As a result, they often cause some degree of high blood pressure (which may require antihypertensive medication) and are associated with an increased risk for kidney damage, bleeding, stroke, and coronary artery blockage. Patients with a history of these or other medical conditions may not be suitable candidates for these drugs.
Your oncologist will discuss the possible benefits and risks of taking any targeted therapy with you. As always, the choice of cancer therapy depends on the optimal treatment for the disease, your health, and your preferences.
Visit www.cancer.net and learn more about managing the side effects of targeted therapies.
Will targeted therapies be tailored to each patient in the future?
Patients want to take drugs that will work for them, and doctors only want to prescribe such drugs. The advent of targeted therapies offered the promise of matching the choice of cancer-fighting drug to measurable characteristics of a patient’s cancer. For example, if drug A targets molecule Z on cancer cells, then those cancers with the most Z should respond the best to drug A. But in what has been the source of great frustration to patients, physicians, and researchers, this simple relationship has not held up for many targeted therapies.
For example, Erbitux binds specifically to EGFR. Yet the amount of EGFR on a person’s cancer cells does not predict how well the cancer will respond to Erbitux. Similarly, Tarceva and Iressa are small molecule inhibitors of EGFR that are used to treat lung cancer; the likelihood of a cancer responding to these drugs is only weakly related to the levels of EGFR present on the cancer cells.
So what does determine the response to a targeted therapy if not the presence of the target? It turns out that for several targets, its structure or shape rather than its absolute amount best determines how a cancer will respond to a targeted therapy. For example, if a lung cancer contains an abnormal (mutant) form of EGFR, then there is a much greater chance that Tarceva or Iressa will shrink the cancer; such mutations are found in only 10 percent of all non-small cell lung cancers, often in those who either never smoked or stopped smoking more than twenty-five years before their diagnosis.
New techniques are emerging that will make it easier to detect overactive communications pathways or mutations in specific molecules in the tumor specimen of each patient. In the future, cancer therapies will increasingly tailor the medicines chosen to the specific molecular alterations contained in any given cancer rather than relying only on the type of cancer a person has.
Are there other kinds of targeted therapies besides those discussed?
You’ve heard the saying, “Build it and they will come.” Well, if a target exists in cancer cells, then scientists will try to target it. Hundreds of targeted therapies are currently being tested. Many will targetcancer cells directly, and others will target the immune system, tumor blood vessels, or the normal organs in which metastatic cancers grow (such as the bone and liver). Some will be specific to one cancer type, others broadly active against a large number of different cancers. Access to some of these new treatments can be found at www.clinicaltrials.gov.
Whenever possible, support cancer research to help bring new therapies to the clinic.