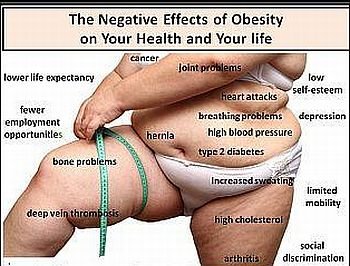
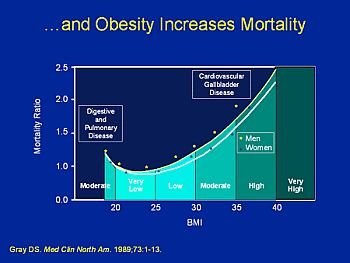
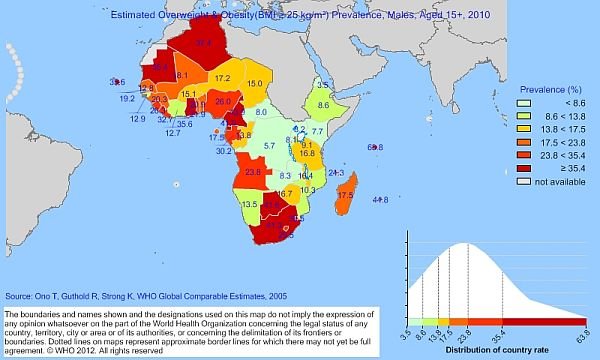
How being overweight or obese causes High Blood Pressure
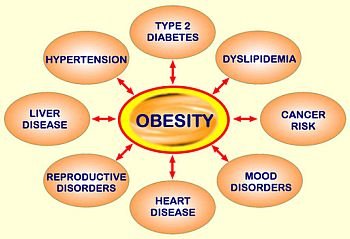
WHY TREAT OVERWEIGHT AND OBESITY?
The recommendation to treat overweight and obesity is based not only on the previously presented evidence that shows overweight is associated with increased morbidity and mortality, but also on randomized controlled trial (RCT) evidence that weight loss reduces risk factors for disease. Thus, weight loss may help control diseases worsened by overweight and obesity and may also decrease the likelihood of developing these diseases. The evidence meeting the panel’s inclusion criteria is presented in the form of evidence statements with a corresponding rationale for each statement. Many of the RCTs examined by the panel included the use of pharmacotherapy for weight loss. When the panel began its deliberations in 1995, two weight loss drugs, fenfluramine and dexfenfluramine, were being used widely for long-term weight loss, i.e., 14 million prescriptions were given out over 11⁄2 years. However, as of September 1997, the Food and Drug Administration (FDA) requested the voluntary withdrawal of these drugs from the market due to a reported association between valvular heart disease and the use of dexfenfluramine and fenfluramine alone or combined with phentermine.
In November 1997, the FDA approved the drug sibutramine hydrochloride monohydrate for the management of obesity, including weight loss and maintenance of weight loss when used in conjunction with a reduced-calorie diet. Thus, at the present time only one weight loss drug is available for long-term weight loss. (Note: FDA approval for orlistat is pending a resolution of labeling issues and results of Phase III trials.)
However, due to rapidly evolving information regarding the use of pharmacotherapy for weight loss, the panel decided to present (below) their critique of those pharmacotherapy trials meeting their criteria for consideration.
Blood Pressure
To evaluate the effect of weight loss on blood pressure and hypertension, 76 articles reporting the results of RCTs were potentially eligible for inclusion. Sixty articles included lifestyle trials that studied diet and/or physical activity, and 16 articles were of pharmacotherapy trials. Dietary interventions included low-calorie diets and diets that promoted macronutrient composition changes, such as amount and type of dietary fat.
Physical activity, when included, was used to help promote increased energy expenditure.
These trials did not always control for other dietary factors that lower blood pressure, such as dietary salt, and the degree to which those taking the blood pressure measurements were blinded to the patient’s change in weight.
Of the 35 lifestyle RCT articles deemed acceptable, 16 included hypertensive patients, and 19 were conducted in individuals with normal or high-normal blood pressure.
Lifestyle Trials in Hypertensive Patients
Evidence Statement: Weight loss produced by lifestyle modifications reduces blood pressure in overweight hypertensive patients.
Evidence Category A. Rationale: A 1987 meta-analysis 381 covering five of the acceptable studies in hypertensive patients concluded that weight loss accomplished by dietary interventions significantly lowered blood pressure. In hypertensive patients, 10 kg (22 lb) of weight loss was associated with an average reduction of 7 mm Hg systolic and 3 mm Hg diastolic blood pressure compared with controls.
Since publication of this meta-analysis in 1987, almost all relevant studies have reported that weight loss reduces blood pressure or the need for medication in hypertensive patients:
The Trial of Antihypertensive Interventions and Management (TAIM), conducted in hypertensive individuals not taking medication for 6 months, reported that, compared with controls, a mean net weight reduction of 4.7 kg (10.4 lb) reduced systolic and diastolic blood pressure by 2.8 mm Hg and 2.5 mm Hg, respectively; the effects on blood pressure were equivalent to drug therapy among those participants who lost 4.5 kg (9.9 lb) or more.
One study in older (age 60 to 80 years) hypertensive adults whose blood pressure medication was withdrawn (Trial of Nonpharmacologic Interventions in the Elderly, TONE) showed that after 2 years, mean net weight loss of nearly 4 kg (9 lb) resulted in more participants free of trial endpoints (occurrence of high blood pressure, resumption of blood pressure medication, or occurrence of a cardiovascular-clinical complication [39.2 percent versus 26.2 percent]).
Furthermore, blood pressure control was similar for men and women and for African-Americans and whites.
In other studies of primarily middle-aged hypertensive adults, compared with controls, weight loss significantly reduced a return to hypertension medication at 1 year and at 4 to 5 years.
The Multiple Risk Factor Intervention Trial, which recruited 12,866 high-risk men, 30 percent of whom had hypertension, delivered an integrated intervention addressing several lifestyle behaviors and included weight loss as an important component. Significant reductions in systolic and diastolic blood pressure were found over 6 years compared with the usual care group and were directly related to weight loss: 1 kg (2.2 lb) of weight loss was associated with a reduction of 0.4 mm Hg systolic and 0.3 mm Hg diastolic blood pressure in men not taking antihypertensive medications. The effect was slightly lower for men taking antihypertensive medications.
Only one RCT conducted in hypertensive patients reported no significant change in blood pressure despite weight loss of 3.3 kg (7.3 lb).
Another study conducted in hypertensive individuals suggested that weight loss reduced blood pressure only when sodium intake was also reduced. This finding was not consistent with a prior study of hypertensive patients and has not been corroborated in subsequent larger trials conducted in nonhypertensive individuals.
Lifestyle Trials in Nonhypertensive Individuals
Evidence Statement: Weight loss produced by lifestyle modifications reduces blood pressure in overweight nonhypertensive individuals.
Evidence Category A. Rationale: A semi quantitative review covering four of the acceptable studies in nonhypertensive individuals concluded that weight loss through dietary interventions significantly lowered blood pressure. In nonhypertensives, excluding one outlier trial that showed very large reductions in blood pressure, 372 1 kg (2.2 lb) of weight loss was associated with a reduction of 0.45 mm Hg in both systolic and diastolic blood pressure. Since this review, published in 1991, almost all relevant studies have reported that weight loss reduces blood pressure in nonhypertensive individuals.
The Trials of Hypertension Prevention Phase 1 (TOHP I) and Phase 2 (TOHP II) are among the larger, well-designed randomized trials, having follow-up rates exceeding 90 percent. They followed the participants for 18 months and 3 to 4 years. The sample size of the weight loss intervention arms ranged from approximately 300 (TOHP I) to 600 (TOHP II). The populations were diverse, consisting of 30 to 35 percent women and 15 to 18 percent African-Americans, and were recruited from 10 centers in TOHP I and 9 centers in TOHP II. Results from both TOHP I and TOHP II demonstrated that weight loss reduced blood pressure and the incidence of hypertension. Compared with controls, in both trials 10 kg (22 lb) of weight loss was associated with a reduction of 7 mm Hg systolic and 5 to 6 mm Hg diastolic blood pressure at 18 months. At 36 months, systolic and diastolic blood pressure was reduced by 6 and 4 mmHg, respectively, for every 10 kg of weight loss.
In addition, weight loss reduced the incidence of hypertension at 18 months by 20 to 50 percent and at 3 years by 19 percent. Secondary analyses from TOHP I and analyses under way from TOHP II demonstrated that the greater the weight loss the greater the blood pressure reduction, and as long as weight loss was maintained, blood pressure remained reduced. One study that focused on older African-American diabetics showed that weight loss of 2.4 kg (5.3 lb) at 6 months resulted in a reduction of 3.9 and 4.0 mm Hg systolic and diastolic blood pressure. Only one study in nonhypertensive individuals showed inconsistent results, where weight reduction decreased systolic (significant) and diastolic (nonsignificant) blood pressure at 12 months in women but not in men. Another study showed short-term (3 months) blood pressure reduction but no longer-term (9 months) blood pressure reduction despite maintenance of weight loss of 9 kg (19.8 lb).
Pharmacotherapy Trials
Evidence Statement: Weight loss produced by most weight loss medications (except for sibutramine) and adjuvant lifestyle modifications is accompanied by reductions in blood pressure.
Evidence Category B. To determine the effects of pharmacological therapy on weight loss and subsequent changes in blood pressure levels, 10 RCT articles were examined. Weight loss studies using pharmacotherapy were conducted predominantly in white women and assumed that dietary changes were uniform in the active drug and placebo groups. All of these studies had a placebo-control group for which dietary recommendations for weight loss were provided; weight loss also occurred in the placebo group. No studies examined whether weight loss using pharmacotherapy results in blood pressure reductions similar to weight loss produced by diet therapy, nor were the results compared with a pure control group with no weight loss. In addition, none of the studies that examined the effect of pharmacotherapy on blood pressure controlled for weight loss. In general, the use of phentermine, fenfluramine, and dexfenfluramine resulted in similar or better weight loss than that seen in the control or placebo group, and reductions in systolic and diastolic blood pressure were concomitantly similar to or better than those observed in the diet plus placebo group. However, one study on dexfenfluramine showed increased blood pressure compared to controls, even though weight loss was greater with dexfenfluramine than with placebo. Another study showed blood pressure lowering consistent with weight reduction produced by dexfenfluramine in women with upper-body obesity, but not in women with lower-body obesity. In a combined summary of data on orlistat presented at he FDA Endocrinologic and Metabolic Drugs Advisory Committee Meeting (held in Bethesda, Maryland, in May 1997), only a small decrease in systolic (approximately 2 mm Hg) and diastolic (approximately 1 mm Hg) blood pressure was observed compared to the placebo. In a study of sibutramine, a net weight loss at 3 months of approximately 2 kg (4.4 lb) did not result in lower blood pressure compared with placebo controls. Eleven double-blind placebo-controlled trials lasting from 12 to 52 weeks showed that sibutramine is associated with mean increases in systolic blood pressure of 1 to 3 mm Hg, in diastolic pressure of 1 to 2 mm Hg, and mean increases in pulse rate of 3 to 5 beats per minute relative to placebo. In hypertensive obese patients, the mean change in blood pressure was the same in the sibutramine group and the placebo group; both dropped. Systolic pressure decreased 5.4 mm Hg in the sibutramine groupand 5.8 mm Hg in the placebo group, while the diastolic drop was 5.9 mm Hg and 3.7 mm Hg, respectively. At the FDA recommended doses, 45 percent of patients had an increase, 35 percent had a decrease, and 20 percent had no change in blood pressure.